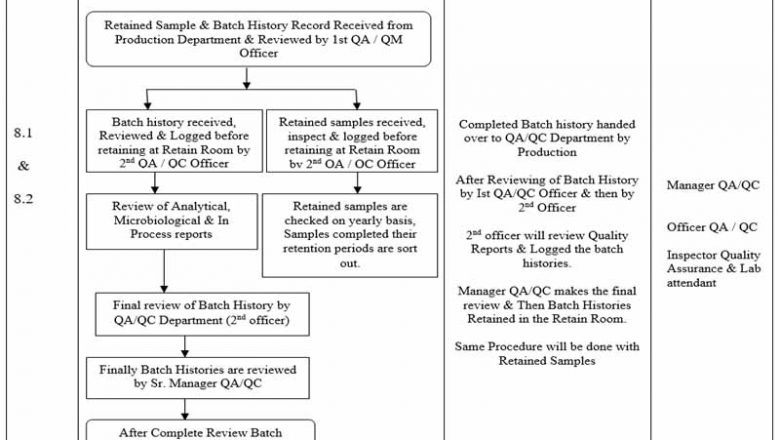
Handling of Retained Samples & Batch History Records
In the post so deeply describe the Handling of Retained Samples & Batch History Records which help Pharmaceuticals in maintaining records.
Objective
To describe the Handling of Retained Samples & Batch History Recordsarticle for receive, review, retention & issuance of Batch History & Retained Sample in compliance of cGMP.
Scope
This procedure is applicable at Pharma.
Responsibilities
Manager QC/QA
Officer Quality Assurance / Quality Management & Quality Control
Inspector Quality Assurance & Lab attendant
Definitions
Batch records
All documents are related to making bulk products or finished products. They provide a history of each batch of product and of all circumstances pertinent to the quality of the final product.
Authorized Person
The pe...




