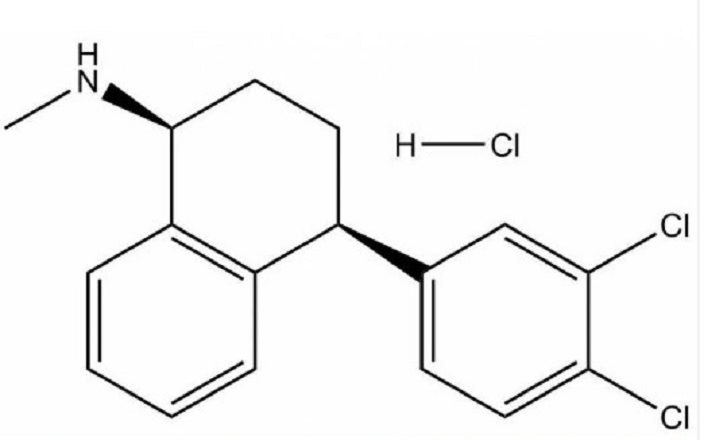
SOP for Finished Product Analysis of Sertraline Hydrochloride Tablet By UV and HPLC Method
Standard Operating Procedure (SOP) for Finished Product Analysis of Sertraline Hydrochloride by UV and HPLC Method
PURPOSE:
To describe the procedure for analysis at the in-process and finished stage of the Sertraline Hydrochloride Tablet.
SCOPE:
This SAP gives a detailed outline for the finished product analysis of the Sertraline Hydrochloride Tablet and will cover In process testing activities on a physical, chemical & instrumental basis.
RESPONSIBILITY:
QC Analyst is responsible for physical/chemical testing and preparing standard analytical procedures.
It is the responsibility of QC Manager to assist and ensure the Testing Procedure as per SAP and to make certain that this SAP is followed in its entirety, reviewed regularly, and revised as necessary.
REFERENCE:
USP 44, NF ...