
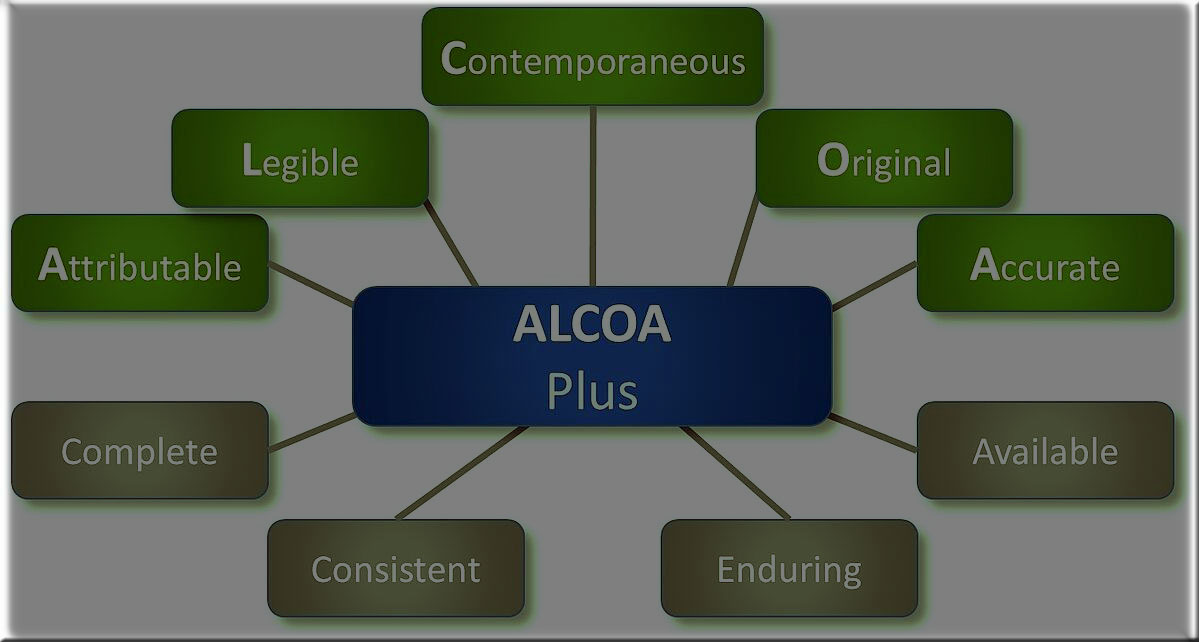
ALCOA+ and Data Integrity in Pharmaceutical is being discussed in this knowledgeable post. Basically, Alcoa is an acronym that stands for Attributable, Legible, Contemporary, Original, and Accurate. Later, Alcoa expanded to Alcoa Plus (Alcoa +), adding a few more concepts that are; Complete, consistent, durable, and available. In addition, the ALCOA + data integrity principles define best practice guidelines and methodologies for good data management, for example, in the pharmaceutical and biotechnology industries. The acronym “ALCOA” defines that the data must be attributable, legible, contemporary, original, and accurate. In addition, the “ALCOA +” guide recommends that the data also be complete, consistent, durable, and available.
With the passage of time, ALCOA+ and Data Integrity in Pharmaceutical concept of Alcoa plus has been adopted by many industries, as the basis for compliance and management of information security and integrity (data protection). Alcoa Plus is used and approved by several major organizations, including the FDA, WHO, and GAMP. ALCOA is above all good information.
ALCOA+ and Data Integrity in Pharmaceutical is an acronym that represents attributes critical to ensuring data integrity in the pharmaceutical industry. The acronym stands for Attributable, Legible, Contemporaneous, Original, and Accurate. Additionally, the “+” sign is sometimes added to emphasize the importance of Complete, Consistent, Enduring, and Available.
Hers is the image for your better understanding of What is ALCOA+ and Data Integrity in Pharmaceutical actually are.
Now let’s we discuss each part of the ALCOA+ and Data Integrity in Pharmaceutical one by one.
1. Attributable
Attributable dictates that any data must be easily identified to the person who performed the data collection, the place of origin and time of data collection must also be noted. In the case of alteration of the data, the person who makes the corrections must also be noted. Ensure that each record is attributable to the person who performed or observed the activity. This involves using unique and traceable user IDs.
2. Legible
Legible data means that the data can be easily read. This attribute must be guaranteed both in the short and long term, so the materials used to record and collect the data must be durable. All records must be clear, readable, and indelible. This applies to both electronic and paper-based records.
3. Contemporaneous
This part of ALCOA+ ensures that the document module meets contemporary activity logging requirements by automatically completing and saving the date and time of each document modification. Record data at the time of observation or performance. Avoid backdating or making entries after the fact.
4. Original
Records must be originals rather than copies or transcripts. Again, this applies primarily to manual record-keeping. For example, you should not write a record on a piece of paper with the intention of completing the main record later, as this can lead to errors. Original data should be captured in the first instance. Avoid relying on copies unless it’s part of a validated and controlled process.
5. Accurate
Accurate means all records must reflect the reality of what happened and must be free of errors. Also, there must be no editing of the original information that results in the loss of that information. Ensure that data is accurate and reflects the actual observations or measurements. Any errors should be corrected promptly, with a clear audit trail ALCOA+ and Data Integrity in Pharmaceutical .
If changes are necessary, those changes should be documented in a way that the original information can be referenced. Nothing should be deleted, blocked or deleted.
6. Available ALCOA+ and Data Integrity in Pharmaceutical
In this part of ALCOA+ and Data Integrity in Pharmaceutical the data must be accessible when needed, during the life of the data. Availability ensures that data meets its use, as it can be applied when the need arises. Ensure that data is readily available for review and inspection by authorized personnel, regulatory authorities, and other stakeholders.
7. Enduring
While this is mentioned in an earlier principle, this ALCOA+ principle places a specific emphasis on ensuring that data is available long after it is recorded, decades in some situations. Data should be secure and unalterable throughout its retention period. It should be protected from loss, destruction, or unauthorized alteration.
8. Consistent
Ensure consistency throughout the data. This includes standardizing formats, units, and methods of recording.
Data Integrity in Pharmaceutical:
Data integrity is critical in the pharmaceutical industry to ensure the safety, efficacy, and quality of products. Failure to maintain data integrity can lead to regulatory issues, product recalls, and compromise patient safety. Some key principles for ensuring data integrity include:
Documentation and Recordkeeping:
Establish robust documentation practices, including the use of logbooks, electronic records, and documentation of all critical activities.
Training:
Ensure that personnel are adequately trained on data integrity principles and understand the importance of ALCOA+.
Quality Systems ALCOA+ and Data Integrity in Pharmaceutical
Implement a robust quality management system that includes regular audits, reviews, and oversight of data integrity practices.
Validation and Qualification:
Validate and qualify systems used for data generation, processing, and storage to ensure they meet predefined requirements.
Audit Trails:
Implement and review audit trails to track any changes made to electronic records. Ensure that the audit trail is secure and cannot be manipulated.
Data Review and Approval:
Implement a system for thorough data review and approval by authorized personnel. This ensures that data is scrutinized for accuracy and compliance.
Data Security:
Implement measures to safeguard data against unauthorized access, alteration, or deletion.
Continuous Improvement:
Regularly assess and improve data integrity processes based on feedback, audits, and emerging best practices.

